Polymer Physics
With few exceptions, the projects undertaken at ARS involve polymers — large molecules built from smaller monomer units. And the properties of the materials we develop depend, first and foremost, on the properties of the polymers that go into their composition.
Practical end products involve the conversion of a liquid state as packaged to a solid state in service. The conversion is the result of (a) a polymer solution being dried (removal of solvent), (b) the growth of finite molecular weight polymers to an infinite polymer network through chemical reaction, or (c) a combination of both processes. Large polymer chains can form entanglements with other chains, and they can condense with themselves and their neighbors under the pressure of van der Waals forces. In some cases, reactive groups may form chemical bonds between adjacent chains. These are examples of crosslinking, by which chains attach themselves to each other, such that force may be transferred from one chain to another. A sufficiently crosslinked system forms an infinite network, a solid, capable of mechanically responding at one location to an input (stress, strain, deformation) at another location, well removed.
Thermoplastics
At low temperatures, the polymer chain segments have little kinetic energy, and are dominated by the attractive van der Waals forces between chains, which lock them together into potential energy minima. They are frozen. In this state the polymer is like other non-elastic solids. Typically amorphous, it is similar to a glass: hard and brittle. Schematically, the material is like the two chains shown below, where parallel segments are frozen together. Although polymer chains are never stretched out straight like shown, this is a convenient way to picture a certain phenomenon. In this state, the polymer network has almost no ability to deform, stretch, or absorb shock. Since it is locked into its configuration, it cannot distribute stress. Rather, stresses are concentrated and borne by relatively few molecular bonds, which cleave under the load, passing stress to neighboring chains, which likewise fail, quickly propagating the crack.
![]()
As the temperature increases, some segments of the network acquire enough energy to break free from their van der Waals bonds and assume vibrational and rotational motion. This early state of thaw is shown below.

Since the unfrozen segments are free to vibrate and rotate, they fold and coil and tend to ball up, much more even than shown in the schematic. This provides the material with the ability to extend itself (deform) in response to a shock or a load, distributing stress among many molecular bonds. And since the temperature is such that some segments have begun to thaw, the input of more energy will cause more of the frozen portion to thaw, lengthening the portion that is unfrozen and in thermal motion, as depicted below. The material is in transition from a glassy to a viscoelastic state.

As the ratio of unfrozen to frozen polymer increases, the material becomes softer and somewhat elastic. The elastic restoring force is a response to the straightening of the vibrating/rotating coiled segments, which is opposed by a reactionary, conservative force that is attempting to maintain the angular momentum of unfrozen chain segments. The viscous character of the material is primarily attributable to the additional thawing of frozen segment, which occurs in response to mechanical strain when the temperature is near the melting point. This is similar to pulling Velcro® apart — it requires work, but the energy is not stored, it is used to change the state of the material from bound to free.
Now if some portion of the frozen polymer is of a different composition from the rest, and if its melting temperature is significantly higher, such as would be the case in a block copolymer like a polyurethane, polyurea, or polyamide, then there is a range of temperatures between the end of the melting transition of the one block and the beginning of the melt transition of the other block. In this region, the material exhibits primarily elastic character. If the usage temperature for the material is in this range, the lower-melting portion is called the soft block; the higher-melting portion the hard block. The soft block provides the elasticity and rubbery character, the hard block holds the network together as an integral solid. And since the soft block is fully melted, viscous forces are essentially absent, resulting in a snappy, resilient material.
If there is no high melting hard segment in the polymer — or if the temperature is increased through the melt transition of the hard segment — and there are no chemical crosslinks between chains, the network comes apart, as depicted below. Chains are completely melted, all segments are in rapid, energetic motion, and the material relaxes and flows as the chains slip past one another. Now the only force resisting deformation is friction between chains (viscosity) and even entangled chains are unraveled as the free volume within the network increases. This is the viscous, or flow, region.
The materials described thus far are thermoplastics. They have no chemical crosslinks, but depend, rather, on the temperature-dependent bonding between chain segments, to hold them together. It is said that they are physically crosslinked. Such materials may be reversibly melted and reformed, or dissolved and re-dried, and represent a very useful class of polymer.
Thermoset Materials
Now if there are branch units distributed along the polymer chain, and these branch units hold reactive groups that chemically bond with branch units on neighboring chains, then the material is chemically crosslinked. It cannot be liquefied by heat or solvent without destructive chemical reaction. It is a thermoset material, as pictured below. Even after all the physical crosslinks have melted, the network remains intact, continuing to exhibit viscoelastic behavior.
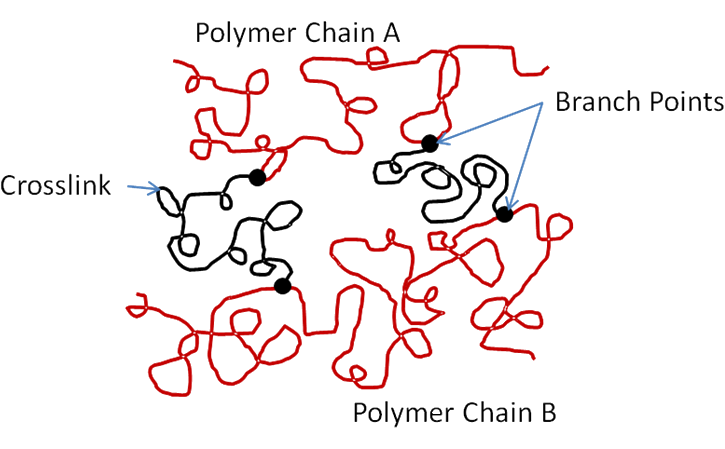
Mechanically, the key quantity of a polymer network that determines behavior is the average size, or molecular weight, of unfrozen effective chain. We distinguish effective chains from ineffective chains. The former have both ends terminating at branch points; the latter have free ends, and can contribute to viscosity, but not elasticity. A branch point is a junction, which could be a frozen block, where three or more chains join. It matters not whether the junction is physical, such as from entanglements or frozen hard blocks, or is a covalent chemical bond. But the number average of all effective chains that bridge between branch points is known as Mc, and is inversely proportional to the effective crosslink density of the polymer network.
Crosslinking and Mechanical Properties
The importance of Mc is that it governs both the stiffness (elastic modulus) of the material and its ultimate elongation. Bigger Mc equates to longer effective chains. Longer chains require less force to stretch by a given amount; and longer chains can stretch greater distances before they are fully extended. Thus as Mc increases, modulus goes down and ultimate elongation goes up. Mc may be increased by raising the temperature — thereby melting some hard block — or reducing the density of chemical crosslinks. It is instructive, at this point to examine a dynamic mechanical analysis (DMA) plot of a polymer material to track the transitions through which a network may go as temperature is increased. In the figure below, the solid trace is the stiffness (elastic or storage modulus) and the dashed trace is the ratio of the loss (viscous) modulus to the storage modulus, known as tan delta. Maxima in the tan delta curve usually indicate phase transitions (melting).
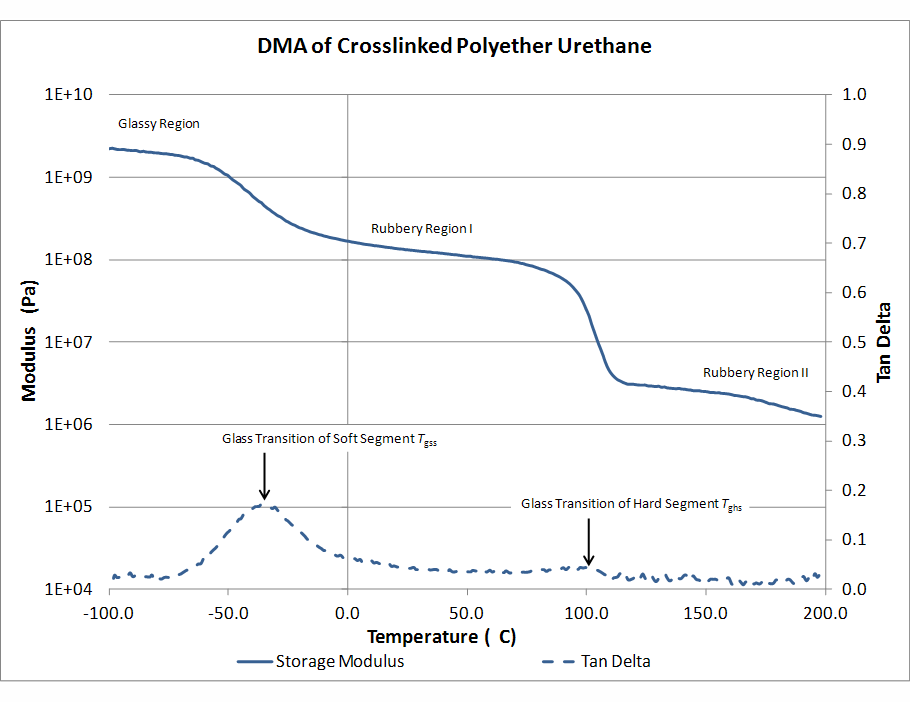
Starting from the far left of this trace, at -100°C, the material is very hard. Tan delta is very low, meaning that there are negligible viscous forces at work. At about -70°C the storage modulus starts to decrease and tan delta starts to rise, indicating the initial thawing of soft segment backbone molecules. As the storage modulus continues to drop through 0°C, Mc is increasing proportionately. This transition has a long tail, which means that some frozen portions continue to melt as the temperature is raised through the rubbery region. From about 20°C to about 80°C the material is mainly elastic, showing little viscous character (tan delta is low). Around 80°C the material starts to soften again and there is a slight uptick in tan delta. Urethane hard segments which are providing much crosslinking are melting and Mc goes up by more than an order of magnitude. From about 120°C and up, the material is being held together almost exclusively by chemical crosslinks, which stem from branch units incorporated into the resin prior to cure. There is another rubbery plateau through which the material is highly elastic, but much softer than it was through the first rubbery plateau. Eventually, excessive temperature will reverse some of the chemical polymerization linkages that comprise the chains and crosslinks and the polymer will irreversibly melt or oxidize.
The transitions through which Mc goes as the temperature changes from low to high provide strong indication of how the material will behave in service. If an application requires elongation of more than 100% at temperatures below –50°C, then the first glass transition should end below about –60°C. The material appearing in the DMA would be too hard and brittle for such an application. If an application requires damping, or the absorption of energy, tan delta should have appreciable value near the temperature of operation. Otherwise, the material will behave too elastically — storing and returning energy — rather than dissipating it. On the other hand, if the material is to serve in a cyclic strain application, such as a motor mount, a high tan delta will cause its temperature to increase as a result of constant cyclic deformation. Care must be taken that the higher temperatures do not lead to melting or other polymer degradation, but also that there is no nearby secondary glass transition that would lead to excessive softening, such as is the case in the DMA presented, around 90°C.
Crosslinking and Chemical Properties
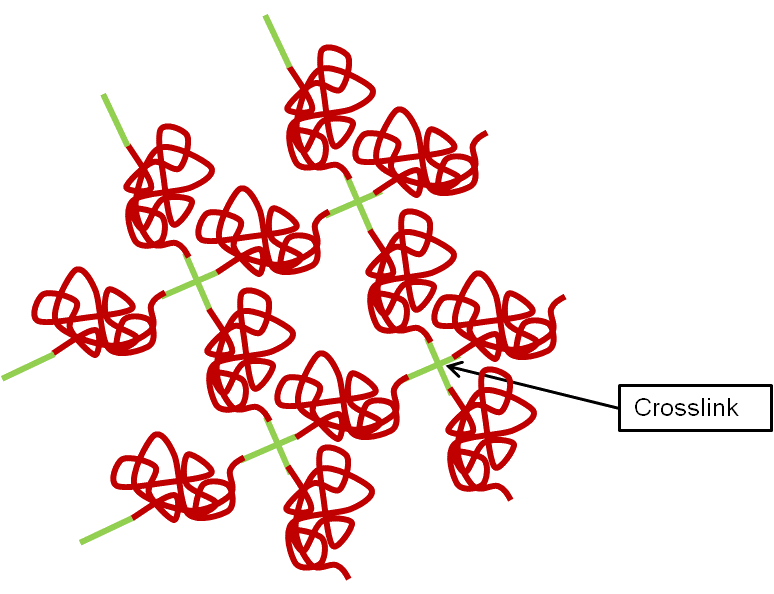
The tendency of polymer materials to swell or resist swelling by fluids and solvents is another important consequence of crosslink density. The natural configuration of a polymer network is one in which the chains are coiled and balled up between crosslinks, as shown simplistically for two dimensions in the figure above. Since physical crosslinks may be melted or dissolved by fluids or solvents, we confine this discussion to thermoset materials, which have chemical crosslinks. Under mechanical stress, the coiled soft segments in the network will extend themselves, up to multiple times their equilibrium end-to-end dimension, depending on their chain length. This is what allows the bulk material to stretch and bend in rubber-like fashion.
If, on the other hand, instead of mechanical stress, the polymer network is exposed to a fluid, then something similar might occur. What ultimately happens depends on two factors: the solubility of the network backbone molecule in the fluid at the temperature of exposure, and Mc, the average length of effective network chain.
When a polymer chain is exposed to a poor solvent, it prefers contact with itself to contact with the solvent molecules; and the solvent molecules feel the same way. Therefore, the polymer chain remains coiled and folded so as to minimize mixing with the incompatible fluid. When the fluid is a good solvent for the network backbone, its molecules prefer contact with the solvent molecules, and they with it. The polymer chain therefore opens up to maximize the mixing and interaction with solvent. In this case, the only factor preventing swelling of the bulk material to several times its equilibrium volume is the shortness of the effective chains. A short chain cannot extend itself, relative to its equilibrium length, nearly as much as can a long chain.
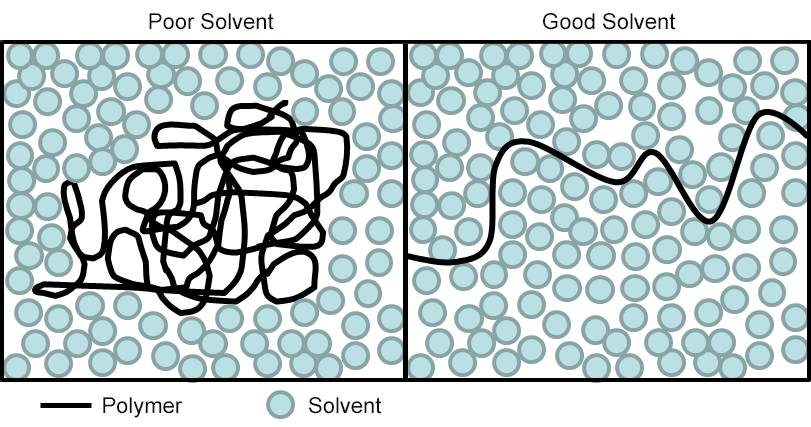
This implies that the only way to minimize swelling in a fluid that is a good solvent for the polymer backbone is to minimize Mc by crosslinking it highly. And, of course, this has implications for the mechanical properties, resulting in a harder, more brittle material that will have less elongation than a more elastomeric variant. Indeed, low fluid swell, high elongation, and low temperature flexibility are difficult properties to obtain in a single material. If high crosslinking cannot be accommodated in a particular case, one must typically resort to alternate backbone molecule types, or to backbone modification through block or graft co-polymerization, whereby molecular segments that are less compatible with the fluid are incorporated into the network structure.
Conclusion
The scientists and engineers at ARS find solutions to many coating, adhesive, sealant, and elastomer problems by first finding the appropriate polymer structure for the requirements of the application. This takes into account temperature, strength, elongation, and flexibility considerations; viscous vs. elastic behavior; dynamics and strain rates; and crosslinking and polymer solubility in fluids common to the service environment. From a solid polymer foundation, additional requirements pertaining to material function, weather resistance, appearance, and durability are far more likely to be met.
